Aptima HPV Test
Aptima HPV Test by Hologic Inc., detects HPV types that belong to a high risk group for causing cervical cancer. Aptima targets mRNA oncogenes Ε6/Ε7 of the virus1. Studies have shown that a test which targets these areas of the virus accurately identify whether there is a risk the infection evolves to cancer 2,3.
Aptima HPV test has been approved by the FDA USA in 2011 for detecting 14 types of high risk HPV and for identifying types 16, 18/45 4.
Sources:
1 Aptima® HPV Assay package insert #503789 Rev A 2013.
2 Tinelli A, et al. Curr Pharm Biotechnol. 2009 Dec;10(8):767-71
3 Cuschieri K, et al. J Med Virol. 2004 May;73(1):65-70
4 Department of Health & Human Services, FDA, P100042, APTIMA HPV Assay
READ MORE
CLART HPV Products
CLART HPV test series detects and at the same time identifies HPV types that are of high, low and medium risk groups. All types are detected in a single trial.
The classification is determined by the innate ability of those viruses to cause cervical cancer or other cancer types (pharyngeal, etc.).
HPV identification makes monitoring women possible, as well as assessing those vaccinated, and it is also useful for post treatment follow-ups.
READ MORE
CLART HPV series consists of the following products:
― CLART HPV 4
Detecting 35 subtypes: High risk (16 18 31 33 35 39 45 51 52 56 58 59 66 68A-68B) possibly high risk (26 53 73 82) and low risk (6 11 40 42 43 44 54 61 62 70 71 72 81 83 84 85 89).
No DNA extraction needed.
Certified for the following types of samples: ThinPrep, Swabs, Biopsies
― CLART HPV 4S
Detecting high risk types (16 18 31 33 35 39 45 51 52 56 58 59 66 68A-68B) and low risk types (6 11).
No DNA extraction needed.
Certified for the following types of samples: ThinPrep, Swabs, Biopsies
― CLART HPV 3
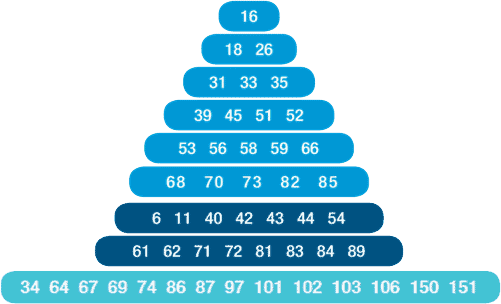
Detecting 49 subtypes: High risk (16 18 31 33 35 39 45 51 52 56 58 59 66 68) low risk (6 11 40 42 43 44 54 61 62 70 71 72 81 83 84 85 89) and not identified risk types (for research only): 34, 64, 67, 69 74, 86, 87, 97, 101, 102, 103, 106, 150, 151.
Suitable for various automated extraction systems
Certified for the following types of samples: ThinPrep, Swabs, Biopsies
HPV molecular testing – The optimal solution
The significance of Molecular Testing
- Comparing to all modern cervical cancer prevention methods, HPV Molecular Testing has the highest negative predictive value in detecting the high-grade CIN1 lesion.
- Higher sensitivity than any other method on CIN2+ lesions (>90%)2,3
- Molecular testing has high reproducibility
- 20% of women diagnosed with cervical cancer had a negative cytology report4
CLART HPV3 – Further advantages
- Application on a large range of samples (thinprep, swabs, specimens fixed in formalin, ethanol or embedded in paraffin)
- Triple test: confirmation a) that DNA is present in the specimen, b) DNA amplification, c) confirmation of virus identification
- Clinical certification 7,8,9
- CE Certified
HPV types identified by CLART HPV3
Sources:
1. Long term predictive values of cytology and human papillomavirus testing in cervical cancer screening: joint European cohort study BMJ 2008; 337:a1754
2. Cervical cancer screening: on the way to a shift from cytology to full molecular screening, M. G. Dijkstra1,2, P. J. F. Snijders1, M. Arbyn, D. C. Rijkaart1, J. Berkhof and C. J. L. M. Meijer
3. Molecular Diagnostics of Human Papillomavirus, Ashley Arney, MT(ASCP)CM,1 Katie M. Bennett, PhD, MB(ASCP), September 2010 Volume 41 Number 9 LABMEDICINE
4. Screening history of women with cervical cancer: a 6-year study in Aarhus, Denmark, O Ingemann-Hansen, M Lidang, I Niemann, J Dinesen, U Baandrup, H Svanholm and L K Petersen, British Journal of Cancer (2008) 98, 1292–1294. doi:10.1038/sj.bjc.6604293
5. Cancer Epidemiol Biomarkers Prev. 2006 May;15(5):908-14.
6. “The Influence of type-specific human papillomavirus infections on the detection of cervical precancer and cancer: A population-based study of opportunistic cervical screening in the united states”. Int J Cancer. 2013 Nov 14. doi: 10.1002/ijc.28605
7. “Clinical Performance of the CLART Human Papillomavirus 2 Assay Compared With the Hybrid Capture 2 Test”. Journal of Medical Virology 83:272-276 (2011)
8. “Prevalence of Human Papillomavirus Infection in Women in Portugal The CLEOPATRE Portugal Study”. Int J Gynecol Cancer 2011;21: 1150Y1158
9. “Patterns of cervical coinfection with multiple human papilloma virus types in a screening population in Denmark”, Goldman B1, Rebolj M, Rygaard C, Preisler S, Ejegod DM, Lynge E, Bonde J. Vaccine, Volume 31, Issue 12, 15 March 2013, Pages 1604-1609, Vaccine. 2013 Mar 15;31(12):1604-9. doi: 10.1016/j.vaccine.2012.12.084. Epub 2013 Jan 10
― CLART HPV 2
Detecting 35 subtypes: High risk (16 18 31 33 35 39 45 51 52 56 58 59 66 68A-68B) possibly high risk (26 53 73 82) and low risk (6 11 40 42 43 44 54 61 62 70 71 72 81 83 84 85 89).
Suitable for various automated extraction systems
Certified for the following types of samples: ThinPrep, Swabs, Biopsies
Sources
1. “Prevalence of HPV Infection among females in the United States”. JAMA. 2007; 297(8):812-81.
2. “High frequency of multiple HPV types in cervical specimens from Danish women”. APMIS 2009, 117: 108-114.
3. “External quality assessment for molecular detection of human papillomaviruses”. Journal of Clinical Virology 48 (2010) 251-254.
4. “Human Papillomavirus 2 Assay Compared With the Hybrid Capture 2 Test”. Journal of Medical Virology 2011 83:272-276 (2011).
5. “Identification of Multiple HPV Types on Spermatozoa from Human Sperm Donors”. PLOS ONE. March 2011, Volume 6, Issue 3, e18095.
Viroactiv HPV test
Viroactiv HPV test detects HPV L1 capsid protein and accurately determines whether a morphology lesion in a woman’s Pap Test may lead to cancer or not.
L1 protein expression is directly associated to precancerous lesions’ evolution to cervical cancer.
The test can be applied to all the cases of Pap test where the cytologist diagnoses ASCUS, AGUS, LSIL, HSIL type morphologic lesions as a prognosis of their evolution.
Sources
1. Correlation of Immunochemical Detection of HPV L1 Capsid Protein in Pap Smears with Regression of High-Risk HPV Positive Mild/Moderate Dysplasia, H. Griesser, U.Schenck, Analytical and Quantitative Cytology and Histology, Oct 2004
2. Immunocytochemical Detection of Human Papillomavirus High Risk Type L1 Capsid Proteins in LSIL and HSIL As compared with Detection of HPV L1 DNA, P.Melsheimer, Acta Cytologica, 2003
The information referred to for general information and informing the public and in any case can not replace a doctor or other suitably qualified health professional. The responsibility of the media carries by ENORASIS SA



